Evo ako se nekome da citat. Na engleskom je al nema veze....
At 31°C (87°F) the less volatile terpenoids start to evaporate, lending the air even more pungent odors.
At 39°C (102°F ) vir tually all of the terpenoids undergo evaporation fairly rapidly.
At 50°C (122°F) THC-Acid decarboxylates as the water molecule held in the carbonate form evaporates. This activates the THC.
At 66°C (150°F) Cannabidiol (CBD) melts and starts to evaporate.
At 185°C (365°F) Cannibinol (CBN) boils.
At 200°C (392°F) THC boils. Clear vapor from a vaporizer.
first of all, bake your pot on a sheet of tin foil at 250-260deg until when you pinch a nug it breaks apart into dust (usualy 10-15 mins for somewhat fresh stuff)
this is the process of converting 80-90% of whatever inactive alkaloids are left into thc and thcv
sometimes it can double or even triple the potency of your bud if you try smoking it after this process... it will bake out most of the smell and flavor though, so if your picky about that and dont care more for the medical sedation, then maybe its not for you. i can make an ounce last much longer this way though. Quote:
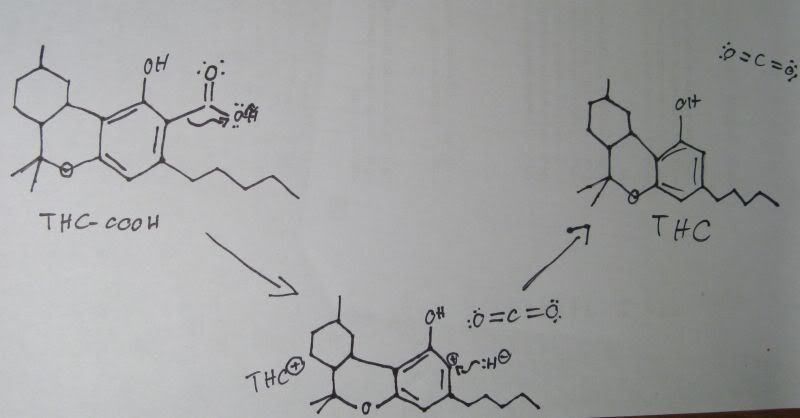
What happens is, a lot of the THC in marijuana is initially in the form of THC-COOH, a carboxylic acid. When heat is applied, a decarboxylation reaction occurs, liberating the carboxyl group as carbon dioxide, with the extra hydrogen atom coming in to bond to the now positively charged carbon atom that was left behind.
Here's my best guess at the actual mechanism:
In the first step, the heat applied excites the molecule and causes the carbon-carbon bond between the benzene ring and the carboxyl group to break and reform on the oxygen molecule in the hydroxyl part of the carboxyl group. This causes the electrons from the O-H bond to move to the hydrogen. There is now a negatively charged hydrogen ion with a lone pair and a carbon dioxide molecule, along with a THC molecule with a positive charge on the member of the benzene ring that the carboxyl group left from.
Once this happens, the hydrogen ion moves in to bond with the carbon in the benzene ring, forming THC. We now have an active THC molecule and a carbon dioxide molecule produced from an biologically inactive THC acid.
I'm guessing that this happens via an SN1 reaction because the carbocation intermediate that is formed is relatively stable due to the positive charge not just being in the benzyllic position, but actually in the benzene ring.
Ovo ne mora biti tocno. Samo spekulacije







 barem tak mislim, nisam bas puno proucavao sve to
barem tak mislim, nisam bas puno proucavao sve to